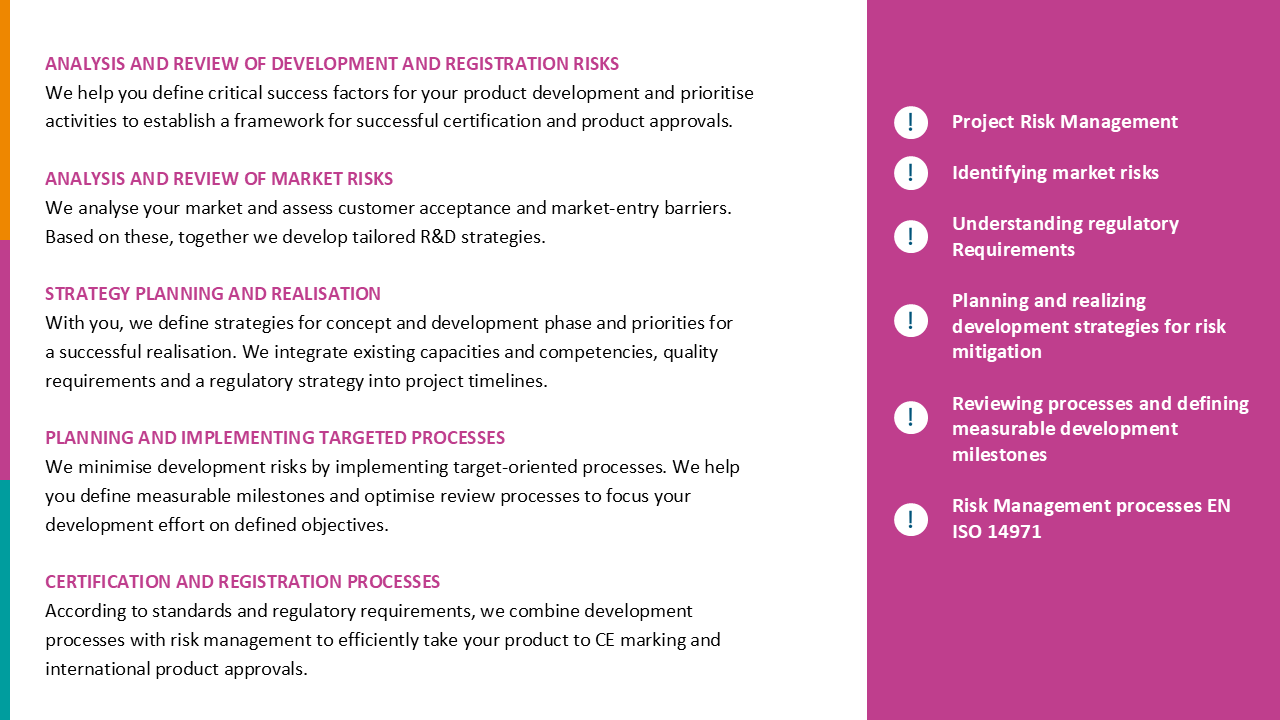
Product Development in MedTech – Pain or Gain?
Whether companies painfully fail in the development of highly complex products or they are successfully approved is primarily a question of routine. If it is lacking, there is a threat of exploding costs, no approval – in the worst case, even the end of the entire company.
In order to lead the development of your innovation to success, know-how, manpower, methods, management skills and tact are required. Or a partner like 3R LifeScience, who accompanies medical technology companies on a daily basis from the product idea to the usable end product.
Our expertise ensures:
- Minimized risks instead of unknown stumbling blocks
- Plannable investments instead of multiplied costs
- Companies on the road to success instead of existential fear
How we were able to help the market leader in the field of pediatric cardiac support and what you can take away from this for your development project?


The biggest risk is not knowing the risks.
Dr. Veit Otto, product development expert at 3R LifeScience, reveals:
“Others call it painting the black – we call it risk management.” Thinking ahead of possible crises and proactively dealing with risks is the most positive thing you can do for your development project. This is the only way to avoid surprises, avoid typical stumbling blocks and set up a resilient project plan.
3 Learnings from development projects in MedTech
In the Berlin Heart project, there were many learnings that contributed decisively to the project’s success. The three most important learnings do not surprise us, because they are typical for complex development projects in medical technology. Perhaps yours as well?
BEST PRACTICE CASE
Develop and approve complex MedTech
3R LifeScience leads Berlin Heart back on the road to success
Who is Berlin Heart?
Berlin Heart develops, manufactures and markets innovative products for mechanical ventricular assist devices (VAD).
It is the only company in the world to provide patients from infants to seniors with reliable systems for VAD therapy.
For more than 20 years, Berlin Heart has been dedicated to the mission of helping people with heart failure live well despite their condition.
With pioneering technologies, the highest precision and the greatest patient orientation, Berlin Heart has established itself from Berlin-Lankwitz as an international market leader in the field of pediatric cardiac support.
A difficult starting position
In April 2015, Dr. Veit Otto from our 3R team of experts stepped in as project manager at Berlin Heart to drive the development of a new mobile, electro-pneumatic drive for the EXCOR® cardiac support system to market readiness.
Previously, the globally respected Berlin-based company had failed to develop this new drive.
In the initial discussions, we identified a number of technical problems: For example, only inadequate product tests had been carried out during development, and in addition to the lack of routine in the development department, there was also a lack of core competencies among the personnel.
But there was also a lot going wrong on the interpersonal level: We perceived strong tensions in communication between the individual departments, and there was a high level of pressure and also frustration among the employees.
To learn more details about the success process managed by 3R LifeScience, which can also benefit your company, simply request our free white paper.
The Result
Success all along the line
CE certification received, FDA approval prepared and project completed on time and on budget – what more could you ask for.
In just four years, Berlin Heart has once again become a viable market leader – with a significant mission, a globally unique product, a qualified team, and further development ideas for a successful future.
Imagine your product successfully seeing the light of day, despite all possible risks and bottlenecks, coming to market and thus giving patients new courage to face life. That’s what we were able to do for the team at Berlin Heart, and now children around the world are benefiting from the result.
Would you like to learn in detail how we achieved this and whether we can do the same for your product?
Berlin Heart says thanks
“When words were followed by actions and actions by results, I was more than convinced! Thanks to 3R LifeScience, we were able to successfully complete more than a singular product development and bring a product to market that has a great impact for our patients.
In addition, we were able to do a great deal of groundwork of an organizational nature within the company, from which we will benefit in the long term.”

Sven-René Friedel
General Manager,
Berlin Heart GmbH
Patients say thanks
And what is your mission that we can support you in?

Product development with 3R LifeScience
When experienced medical technology experts are at work, you can count on clarity and cost control. You can be sure that the development process is adapted to your company’s resources and that you create an approvable end product.
Concentrate fully on your strength: your product. With our know-how, we provide you with the necessary framework conditions and how they can be met in production.
Download
Whitepaper on product development in MedTech
With the free whitepaper, you will receive the complete best practice case Berlin Heart as well as a project risk checklist as a PDF by e-mail. In addition, we will be happy to send you further useful assistance for your development project. [ONLY AVAILABLE IN GERMAN RIGHT NOW)