Never lose track of your Technical Documentation
Benefit from experienced process engineers and medical device experts
Do you have difficulties collating your Technical Documentation in a structured way and keeping it up to date?
Is it time-consuming to collect all the information from various departments in a structured way?
Do you quickly lose track of large amounts of data and complex Excel spreadsheets?
Are you struggling with off-the-shelf IT tools that are supposed to give you an overview but do not fit your company´s structures and processes?
Do you lack the necessary documents to update the Technical Documentation in the event of product changes?
At 3R LifeScience, we ensure that you always have an overview of the technical documents despite the increasing complexity of projects.
Together with you, we create a digital solution that contains the key processes and data lists to create the Technical Documentation initially and update it efficiently in the event of changes.
You want to prove that your products fulfil the technical and regulatory requirements for safety and performance.
You want to make the Technical Documentation readable and comprehensible to third parties, so reviews become easier.
You want your product life cycle data to be available in a structured way and remain always up to date.
You want to make your product knowledge available to new product generations.
You want to be able to react to changes at any time and efficiently integrate and maintain them in the Technical Documentation.
Use our process and development expertise to create a customised digital solution that helps you build and maintain your Technical Documentation.
We offer an innovative solution for setting up and organising your Technical Documentation. In doing so, we follow the structure of the Technical Documentation MDR 2017/745 Annex II/III.
With our approach, you link data and documents to create a comprehensive and well-organised information architecture you can maintain.
Thanks to interactive dashboards, you always keep an eye on the status and important metadata of your documents.


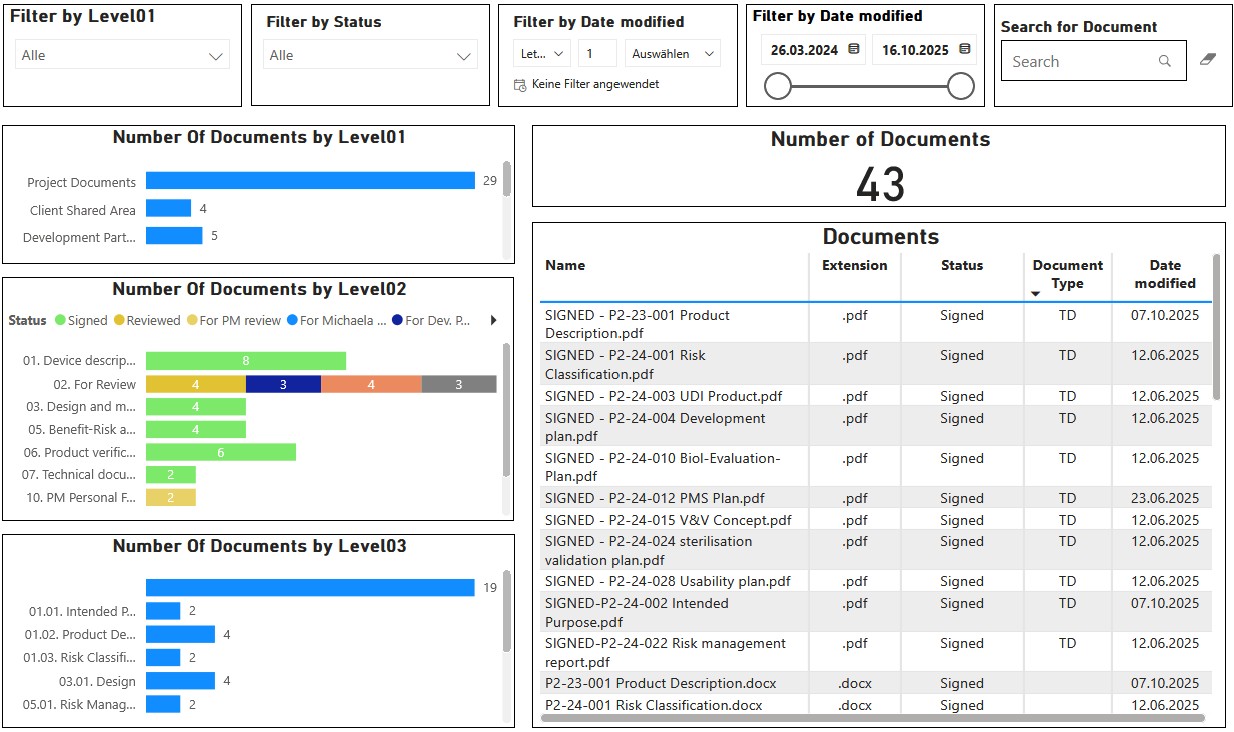
In addition to structuring a Technical Documentation, our tools offer further advantages to your organization.
Other company-relevant processes can be mapped. Relevant dashboards help monitoring critical processes, such as
- Requirements management, GSPR (MDR 2017/745 Annex I) and technical standards
- Development planning
- Risk Management
- Technical Documentation gap analysis and remediation planning
Our solutions reflect many years of experience in medical device development and integrate relevant regulatory requirements. They can be tailored to match your products and your organisational infrastructure.