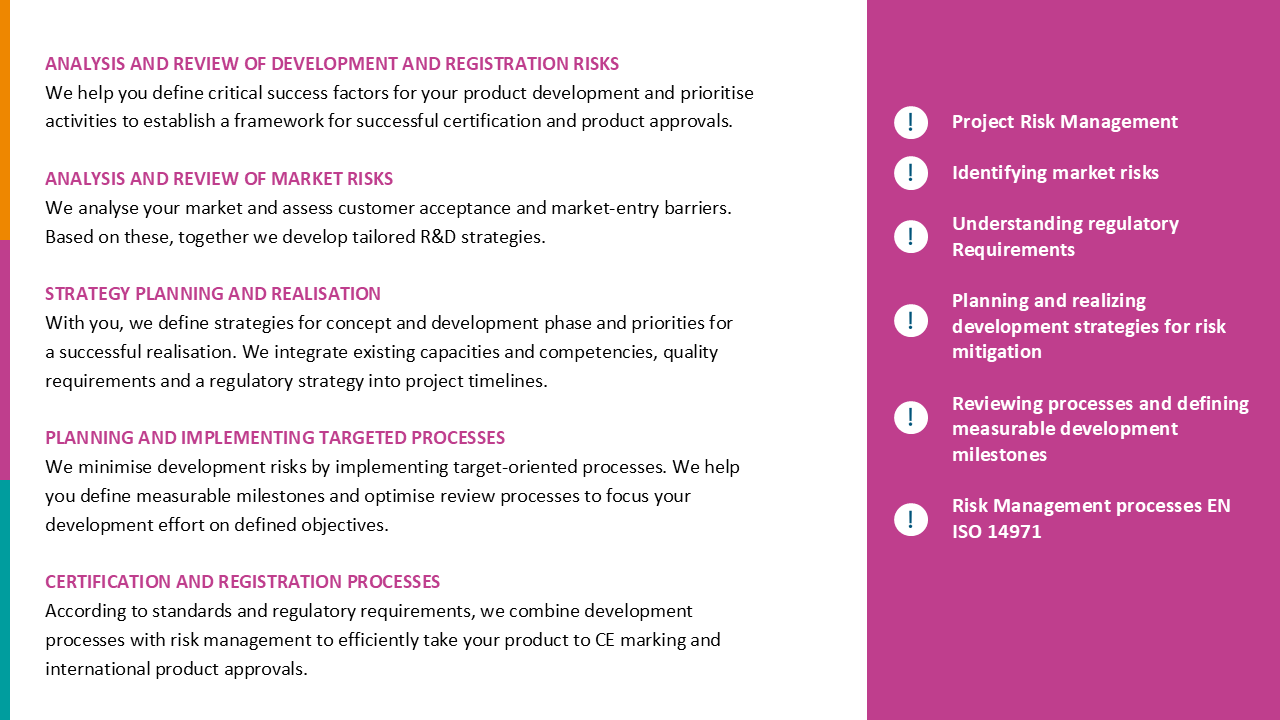
What challenges do you face in developing innovations?
 Customers, Markets and Product Requirements
Customers, Markets and Product Requirements
-
- Knowing clinical needs and requirements
- Developing product specifications
- Taking into consideration standards and regulations
 R&D Concepts and Development Plans
R&D Concepts and Development Plans
-
- Intended Purpose, classifications and borderline
- Risk-based project planning and budgets
- Implementing GSPR and TD requirements
 Technical Specifications and Material Selection
Technical Specifications and Material Selection
-
- Components that meet medtech requirements
- Materials with defined chemical, physical and biological properties
 Design Input/Design Output und Design Control
Design Input/Design Output und Design Control
-
- Design stages, milestone reviews and efficient and compliant approval
 Transfer to Production and Scale Up
Transfer to Production and Scale Up
-
- How to plan production? What to communicate?
- What to test? What to validate?
 Medical Device Verification & Validation
Medical Device Verification & Validation
-
- Biological Safety and Biocompatibility
- Cleanliness and Hygiene in Production
- Usability and Human Factor Engineering
- Packaging, Transport and Shelf-Life Studies
- Electrical and Electronic Safety
- Cybersecurity
- System Compatibility
 Clinical Evaluation and Clinical Studies
Clinical Evaluation and Clinical Studies
-
- CEP & CER DIS/ISO 18969
- Clinical evidence and equivalence to legacy products
- ISO 14155 Clinical Investigations
- Clinical development strategies
- Clinical risks and mitigations
 Change and Product Life Cycle Management
Change and Product Life Cycle Management
-
- Design control
- Product and process changes
- Life time and discontinuation
 PMS and Vigilance Processes
PMS and Vigilance Processes
-
- PMS and Vigilance concepts
- Vigilance reporting forms
- Integration in a Risk Management System
At 3R LifeScience, we ensure that you can implement your product ideas efficiently and in compliance with European and international legislation addressing increasing complexity with smart approaches.
With us product development becomes more planable
Your goal is to develop products as efficiently as possible and in compliance with the law.
You want to avoid delays, cost explosions and other unpleasant surprises in development, verification and approval
You want to sustainably optimize R&D processes throughout the company.
You want to create commercially successful products that are appreciated by patients and users.
You are willing to bring strategic development or technology partners on board to compensate for competence and capacity bottlenecks.
Use experienced medical devices project managers for efficient and transparent R&D activities.
From possibility to reality
From requirements management to the development of feasibility studies, functional samples and prototypes to verification, validation, approval and market launch – our specialized project managers help you at any project phase.
These companies we have successfully supported:




Case Studies
Project management development of a class IIb/ III product
The company: Internationally active medical technology company in the field of orthopedic implants and instruments.
The task: Review and set-up of processes for CE marking and product registration of medical devices.
Our work: We evaluated the status quo of processes for CE marking and product registration of class I, IIa, and IIb medical devices and implemented additional processes to ensure that regulatory requirements for conformity assessment of both national and international registrations are met. This involved optimizing Regulatory Affairs’ interfaces with other departments and taking steps to integrate the new processes and structures into the company’s quality management system.
Customer benefits: Targeted measures created structures and processes that enabled our customer to optimize its internal processes for CE marking and improve communication with Notified Bodies and national registration authorities. The sustainability of our measures was confirmed in audits.
Product portfolio recertification and MDR gap analysis
The company: Internationally active medical technology company with resorbable bone substitute materials.
The task: Planning and execution of the recertification of the product portfolio according to the European Directive as well as gap analysis regarding MDR requirements.
Our work: We reviewed the technical documentation for the Class III product portfolio and created a new design dossier as part of the recertification. The new MDR requirements and requirements from technical standards were defined and implemented in consultation with the Notified Body.
The customer benefit: Our client’s product portfolio was recertified on time and within budget. New regulated requirements were reviewed and comprehensive remediation measures were initiated to further improve the documentation of the established product portfolio.
“When words were followed by actions and actions by results, I was more than convinced! Thanks to 3R LifeScience, we were able to successfully complete more than a singular product development and bring a product to market that has a great impact for our patients.
In addition, we were able to do a great deal of groundwork of an organizational nature within the company, from which we will benefit in the long term.”

Sven-René Friedel
General Manager,
Berlin Heart GmbH
“We understand that the new medical device regulations affect many organisations and raise many questions.”
Together with one of our experts, you clarify your requirements and questions. If cooperation appears helpful, we will arrange a workshop with you.
If you already have clarity about your project requirements and now need support as soon as possible, do not hesitate to call us.