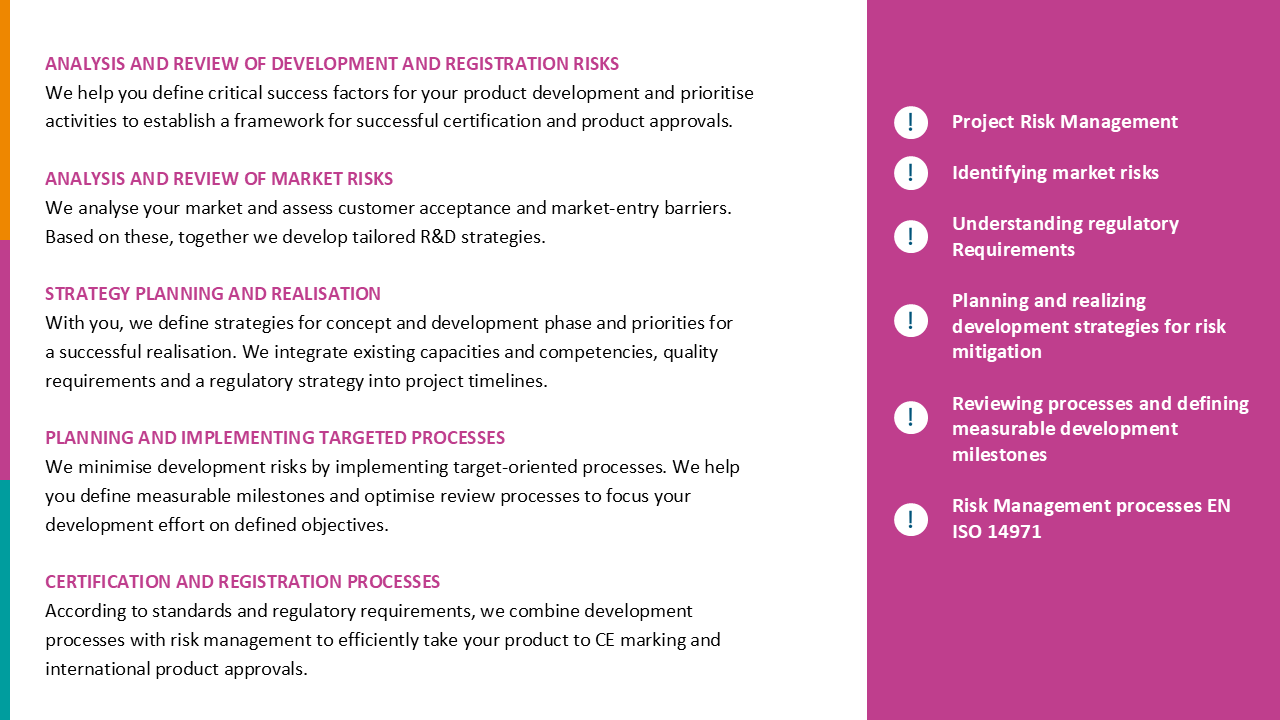
Do quality management and product approval processes challenge your organisation?
QMS according ISO 13485, internal and external Audits
CAPA, process improvements and KPIs
Internal and external interface management
MDR/IVDR, 21 CFR 820/QMSR, MDSAP requirements
MDR/IVDR Technical Documentation, GSPRs, international registrations and approvals, communication with Authorities
At 3R LifeScience, we ensure that you regain control of the growing regulatory and legal complexity in your company.
With us, quality management becomes a basis for efficient and compliant organisational processes
You want all departments involved in QM to communicate and work effectively with each other to contribute to the company’s success.
You want to face the next ISO 13485 audit in a focused an structured way with good processes and documentation
You want to make sure that your products always meet current regulatory requirements.
You want to establish clear QM processes and structures in your company so the next change in legislation can be implemented efficiently.
They want to bring together business and regulatory strategies so all functional units in the company work towards a common goal.
Quality management system according to EN ISO 13485 – your path to certification
The current version of EN ISO 13485 harmonized with MDR/IVDR requirements calls for an even stronger risk-based approach to the development, production, distribution and follow-up of medical devices than before.
Together with you, we create and optimise the corresponding infrastructure, document procedures and methods. We evaluate the results of statistics, audits and monitoring measures and ensure the further development of the QM system based on the applicable standards and regulatory requirements.
This way, you are perfectly prepared for the next audit and reduce your QM overhead at the same time.
We check your “MDR readiness”
Do you want to fulfil the MDR requirements on time?
In this case you must meet the conditions for the application of the extended transitional periods and have these dates clearly in mind!

We ensure that you fulfil all requirements on time and can comply with the new transition periods.
Your path to MDR compliance
- We conduct GAP analyses and actively accompany you to achieve MDR compliance of your processes and products.
- We analyse where you are on the path to MDR compliance and define necessary corrective measures and coordinate their implementation.
- We review all processes for development, production, placing on the market and follow-up.
- We work with a risk-based approach and integrate risk management aspects into all your processes and product developments.
- We support you in establishing and continuously reviewing quality assurance processes
Focal points for MDR implementation
- Re-classification
- Technical documentation
- Verification and validation
- Sterilisation, packaging and labelling
- Clinical evaluation and equivalence assessment (MEDDEV 2.7/1 Rev. 4)
- Post-market clinical follow-up (PMCF)
- PMS and Vigilance
- Supplier Management
- OEM and PLM contracts
- UDI
MDR compliance at suppliers and external service providers
The MDR explicitly requires documented control over suppliers and service providers (such as packaging, sterilisation, warehousing). For this reason, we independently audit your supplier management for quality assurance and control. We analyse your goods-in process and documentation of supplied raw materials, semi-finished products and services in the legal framework of the MDR.
Approval outside Europe
We support you, for example, with FDA approval and develop registration strategies for your CE-marked product in selected markets outside the EU.
Through an established network of partners, we support your entry into selected international markets. This includes communication with authorities, country representatives and independent local licence holders.
Trust the know-how of our QM experts to help your quality management to improve.
From possibility to reality
From requirements management to the development of feasibility studies, functional samples and prototypes to verification, validation, approval and market launch – our specialized project managers are at your disposal for any project phase.
These companies we have successfully supported:




Case Studies
Project management development of a class IIb/ III product
The company: Internationally active MedTech company in the field of orthopedic implants and instruments.
The task: Review and set-up of processes for CE marking and product registration of medical devices.
Our work: We evaluated the status quo of processes for CE marking and product registration of class I, IIa, and IIb medical devices and implemented additional processes to ensure that regulatory requirements for conformity assessment of both national and international registrations are met. This involved optimizing Regulatory Affairs’ interfaces with other departments and taking steps to integrate the new processes and structures into the company’s quality management system.
Customer benefits: Targeted measures created structures and processes that enabled our customer to optimize its internal processes for CE marking and improve communication with Notified Bodies and national registration authorities. The sustainability of our measures was confirmed in audits.
Product portfolio recertification and MDR gap analysis
The company: Internationally active MedTech company with resorbable bone substitute materials.
The task: Planning and execution of the recertification of the product portfolio according to the European Directive as well as gap analysis regarding MDR requirements.
Our work: We reviewed the technical documentation for the Class III product portfolio and created a new design dossier as part of the recertification. The new MDR requirements and requirements from technical standards were defined and implemented in consultation with the Notified Body.
The customer benefit: Our client’s product portfolio was recertified on time and within budget. New regulated requirements were reviewed and comprehensive remediation measures were initiated to further improve the documentation of the established product portfolio.
“When words were followed by actions and actions by results, I was more than convinced! Thanks to 3R LifeScience, we were able to successfully complete more than a singular product development and bring a product to market that has a great impact for our patients.
In addition, we were able to do a great deal of groundwork of an organizational nature within the company, from which we will benefit in the long term.”

Sven-René Friedel
General Manager,
Berlin Heart GmbH
“We understand that the new medical device regulations affect many organisations and raise many questions.”
Together with one of our experts, you clarify your requirements and questions. If cooperation appears helpful, we will arrange a workshop with you.
If you already have clarity about your project requirements and now need support as soon as possible, do not hesitate to call us.
MedTech Services
Follow us for the latest updates from the MedTech world:
Homepage | Imprint | Privacy Policy
© 2026 LifeScience GmbH